 Nucleotide Excision Repair
Nucleotide Excision Repair
Nucleotide Excision Repair (NER) is an important DNA repair mechanism able to remove a broad range of different types of helix-distorting DNA lesions. NER protects organisms against DNA damage-induced carcinogenesis and premature aging. Its significance is illustrated by the severe clinical consequences associated with inherited defects in NER. Genetic defects in NER give rise to various photo-sensitive syndromes, which include the cancer-prone disease Xeroderma Pigmentosum (XP), the premature ageing Cockayne’s Syndrome (CS), the brittle hair disorder Trichothiodystrophy (TTD) and the UV-sensitive syndrome (UVSS). The underlying genetic and molecular defect of UVSSA was recently elucidated by us and others (Schwertman et al, Nat Gen, 2012).
Mechanism
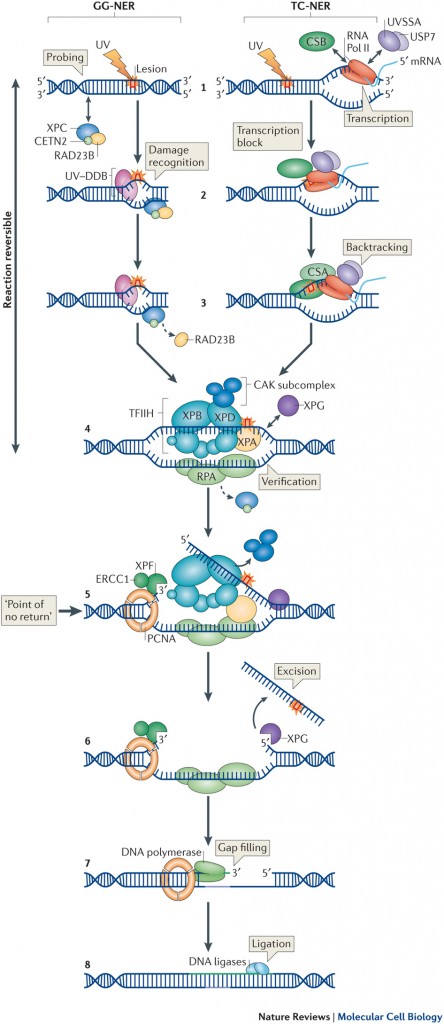
NER is initiated by two distinct DNA damage recognition mechanisms:
(1) In Global genome NER (GG-NER) DNA lesions located anywhere in the genome are detected by the combined activity of the UV-DDB and XPC complexes
(2) In Transcription-coupled NER (TC-NER) specifically DNA lesions located in the transcribed strand of active genes are detected by lesion-stalled RNA polymerase II, followed by the recruitment of the CSA, CSB and UVSSA proteins.
After lesion detection, either via GG-NER or TC-NER, the TFIIH complex is recruited to open the DNA helix and verify the presence of the damage. Next, XPA and RPA stabilize the repair complex and properly orient the structure specific endonucleases ERCC1/XPF and XPG to excise the damaged strand. The resulting ~30 nt single strand DNA gap is filled by DNA synthesis and ligation.
The mechanism of NER and the impact of defects in this pathway on cancer and aging are described in detail in our recent comprehensive review entitled Understanding nucleotide excision repair and its roles in cancer and ageing.
Research in the lab
We apply a multidisciplinary approach to improve our understanding of the molecular mechanism of NER and the impact of NER on biological systems, working from the molecular genetic, biochemical and cell biological level to the level of intact eukaryotic organisms and patients. Research in the lab is focused on:
Mechanism and function of NER
We study the mechanism and function of the individual NER proteins in mammalian cells, by live cell imaging (see the movie for an example of a repair protein that localizes to DNA damage), genetic, biochemical and proteomic approaches. Furthermore, the function of proteins and their impact on health is studied in model organisms such as mice and C. elegans.
Regulation of NER and interplay with other processes
We are particularly interested in understanding how NER functions and how it is regulated within the in vivo environment of the nucleus. Research questions include how NER is controlled by posttranslational modifications, in particular by the ubiquitin proteasome system, and how NER operates within chromatin and how NER is coordinated with other processes in the cell such as transcription.